The electronic configuration of carbon is 1s 2 2s 2 2p 2. However, in some chemical reactions, carbon can lose or gain electrons, which can result in a positively or negatively charged ion.Īlso Read: Structure of an Atom 3. In a stable carbon atom, the number of protons (positive charges) is equal to the number of electrons (negative charges), which results in a neutral charge for the atom. The atomic number of carbon is 6, which means that a neutral carbon atom has 6 protons and 6 electrons. 
The next electron must move to the next shell when one shell is full.Ī neutral carbon atom has 6 electrons. 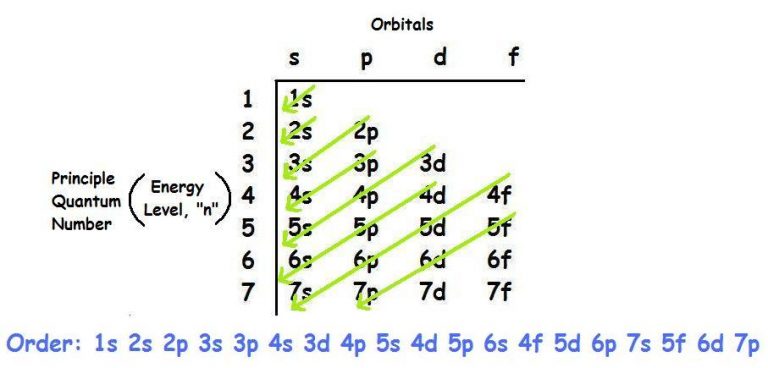
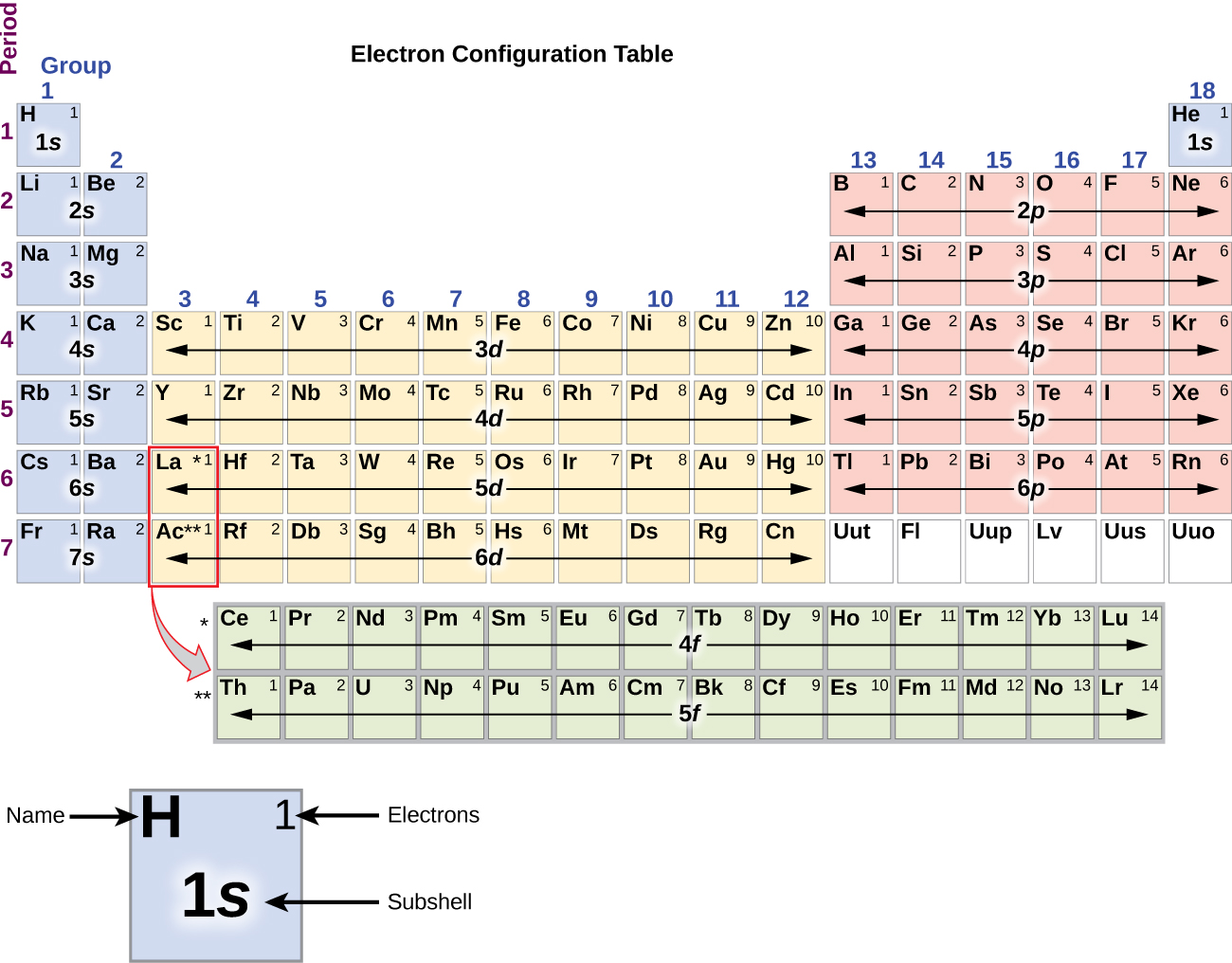
Shell three can hold more than eight electrons, as you come across elements with more than eighteen electrons. Shell number one can only hold two electrons, shell number two can hold eight, and shell number three can hold up to eight electrons for the first eighteen elements. The electrons prefer to be in different shells/orbitals. Number of Electrons in CarbonĮlectrons in an atom spin around the center, also known as the nucleus. It is also a major component of many minerals and fuels, including coal, oil, and natural gas.Ĭarbon has a variety of physical and chemical properties, including its ability to exist in multiple allotropes, such as diamond, graphite, and fullerenes, which have different properties and applications. It is a non-metallic element that is essential to all known forms of life, as it is a key component of organic molecules such as proteins, DNA, and carbohydrates.Ĭarbon is unique in its ability to form long chains and complex structures, which allows it to participate in an almost infinite number of chemical reactions. 1999‑2023 - All Rights Reserved.Carbon is a chemical element and has the chemical symbol “C” and atomic number 6. Īlso see: The Orbitron: A gallery of atomic orbitals and a few molecular orbitalsĬopyright © Israel Science and Technology Directory. The full story of the electron configurations of the transition elements. Thus, each proton and neutron has a mass of about 1 amu. This isotope of carbon has 6 protons and 6 neutrons. Atomic mass is measured in Atomic Mass Units (amu) which are scaled relative to carbon, 12C, that is taken as a standard element with an atomic mass of 12. Each element is uniquely defined by its atomic number.Ītomic mass: The mass of an atom is primarily determined by the number of protons and neutrons in its nucleus. Atomic number: The number of protons in an atom. The table below shows the full forms of the electron configurations of noble gases. For an explanation of these aspects, see the reference by Schwarz listed below. Thus, the electron configuration of Sc is 3d 1 4s 2. Starting with Scandium (Sc, atomic #21), the 3d orbital has a lower energy than the 4s. Energy levels and sublevels Principal energy level Therefore, orbital 4s is filled with electrons prior to orbital 3d. The reason for this is that the energy level of orbital 4s is slightly lower than that of orbital 3d. Note that in the electron configuration of both K and Ca, the 4s orbital is filled before the 3d orbital. The one additional electron configuration completes the picture for 19 electrons of Potassium. The abbreviated form - 4s 1 - means the electron configuration of Argon (Ar), plus one electron in the 4s orbital. The full electron configuration of Potassium (K) is 1s 22s 22p 63s 23p 64s 1. Thus, the configuration shown for Potassium is 4s 1 (see Table below). 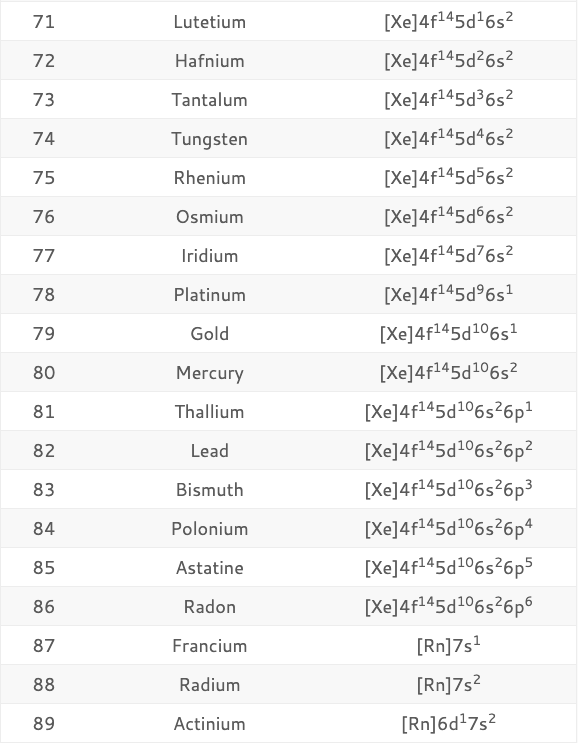
Thus, substituting the config of He gives the full config for Neon: 1s 22s 22p 6įor example, for Potassium (K) (atomic #19), the preceding noble gas is Argon (Ar) (atomic #18). For example, the abbreviated configuration for Neon is 2s 2 2p 6. In the periodic table beyond Helium (He), each element's electron configuration is shown in an abbreviated form that starts with the symbol of the noble gas that precedes it. The superscript shows that there is one electron in the 1s orbital. The simplest configuration is for Hydrogen: 1s 1. An atom's electron configuration describes the distribution of its electrons in the atomic orbitals ordered by the orbitals' energy levels.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
